Open Access Journal of Dental and Oral Surgery
[ ISSN : 2833-0994 ]
Derived Dentin Graft as Autologous Bone Regeneration in Implant Surgery: A Histological and Radiographic Investigation
1Department of Medicine and Surgery, University of Milano-Bicocca, Milan, Italy
2University of Milano-Bicocca, Milan, Italy
3Student, University of Milano-Bicocca, Milan, Italy
Corresponding Authors
Keywords
Abstract
Background: This study has been conducted to evaluate the dentin regeneration potential as an autologous graft material for the preservation of the bone volume in the post-extraction socket. Tooth Transformer has been used as the device required to demineralize the dentin got from the extracted elements, which led to obtaining a granular material rich in collagen and growth factors.
Methods: All 8 patients selected for the study underwent two surgical procedures. During the first surgery, the affected elements have been extracted, the Tooth Transformer protocol has been run, and the material has been grafted into the existing bone defects and post-extraction socket. After three months, the biopsy of the regenerated areas has been performed and the implants required for prosthetic rehabilitation were placed in the same site. For two of the 8 patients, the implant has been placed with the support of a surgical guide.
Result: CBCT and histological analysis’ results after 3 months confirmed the actual replacement of grafted material and the regeneration of bone tissue suitable for implant rehabilitation.
Conclusion: Thus, the Tooth Transformer proves to be an effective device to produce an innovative autologous material from dentin, which is capable of predictable viable bone tissue regeneration able to support prosthetic implant rehabilitation in edentulous sites.
Introduction
Over the past ten years the volume of patients needing dental rehabilitation supported by implants has increased significantly since it has been scientifically proven that a fixed dentition improves the quality of life. However, on patients suffering from periodontal disease or previous edentulousness of one or more teeth, the occurrence of bone defects preventing implants insertion is frequently detected [1-3]. Therefore, bone regeneration is required. Nowadays, a significant number of scientifically validated techniques are available to bone regeneration purposes. All these techniques involve the use of materials from autologous, homologous, heterologous or xenographic origin [4,6]. Autologous-derived material has always been considered the gold standard for bone regeneration, due to its full biocompatibility, richness in growth factors, and highly replacement rate [7]. On the other hand, autologous materials, have some disadvantages. They absorb quickly and must be taken from the same area as the surgery or from another area purposely created. For these reasons, in the last 10 years, have been developed many different biomaterials, with rapid or slow reabsorption. They used as scaffolds because they show osteoconductive properties, which is crucial in the regeneration [8,9].
Osteoconduction is the property that a material possesses to be able to act as a scaffold, where the blood clot can be organised, which will later be transformed into new bone tissue by metaplasia. The best known osteoconductive materials are xenographic, such as coral and animal bone, and alloplastic materials, such as hydroxyapatite, TCP, bioglass and derivatives of polyglycolic and polylactic acid. Because dentin obtained from extracted compromised elements, avoids collection of material from another donor site, its usage for bone regeneration is recommended [5,18]. Idea of use dentin for bone regeneration origins from the fact that ratio of organic material and inorganic material are nearly the same both in dentin and alveolar bone [10-12]. In details, excluding water, their organic matrix consists of 90% collagen and 10% in other proteins which includes many growth factors, while other studies shown that enamel behaves in regeneration phase as a material of heterologous origin or xenographic origin. In 1967, Bang et al. showed the osteoinduction potential of demineralized dentin matrix [13,14]. In 1991, Bessho et al., in an animal trial, detected bone morphogenetic proteins (BMP) after a demineralization process of dentin [15]. In 2017, Rijal et al- elaborated a theory that illustrates a better bone increase due to a higher BMP concentration [16]. In 2021 Minetti et al confirmed the potential role of dentin used in the maintenance, preservation and augmentation of bone tissue given high stability, low level of bone loss around dental implant and high survival rate of the dental implants one year after prosthetic loading [5,17,18].
Materials and Methods
The purpose of the study is to evaluate the regenerative potential of dentin, derived from the dental elements now compromised and extracted, previously treated using Tooth Transformer device, and subsequently grafted in postextraction socket or in sites that already have a bone defect. After 3 months from first surgical procedure, a sample is taken from the same area where the graft was placed and then one or more osseointegrated implants are inserted, depending on the type of rehabilitation chosen for the patient. The analysis of the results is performed both using radiographic examinations (intraoral radiography, orthopantomography and Cone Beam CT) before and after regenerative surgery, before and after biopsy and implant surgery, and in months and years following prosthetic loading, and through histological analysis under an optical microscope. In the case of radiographic examinations, the quantity and quality of the bone tissue gained after regeneration and the degree of resorption over time following the load dictated by the prosthesis will be evaluated. In the histological analysis, however, the presence or absence of grafted material granules, the presence of osteoblasts and osteoclasts and proteins that act as osteoinductive factors, and the presence of new mineralized bone tissue or in the mineralization phase will be evaluated.
Inclusion criteria
The inclusion criteria were age over 18, stable health conditions (ASA-1 and ASA2), presence of one or more compromised teeth, therefore, to be extracted, acceptance of an implant-prosthetic treatment, informed consent and acceptance of the treatment plan using a fixed prosthesis supported by implants
Exclusion criteria
The exclusion criteria were insufficient oral hygiene, a heavy smoker, alcohol or drug abuse, odontostomatological infections, remote or recent radiotherapy in the oromaxillofacial area, recent chemotherapy, recent bisphophon therapy, state of pregnancy, bone grafting required and uncontrolled diabetes mellitus. The conditions present in the exclusion criteria all concern an increase in the probability of not having proper healing and therefore a failure both in terms of the integration of the graft in recipient site and for the correct integration of the implant.
Extractive and regenerative surgery
Antibiotic therapy (1 g of amoxicillin/clavulanic acid in one solution twelve hour and an hour before the surgery) was administer. The compromised teeth extraction and regenerative procedure were all performed using only plexus anesthesia injecting Mepivacaine with the addition of adrenaline (concentration 1:100000). Simple extractions were performed to extract all dental elements, therefore osteotomy and / or odontotomy were not required. No complications were shown during the surgery: bleeding remained within the limits and patient was alert during each phase. After extraction, dental elements were cleaned with physiological solution and any inflammatory and soft tissue, periodontal ligament and other residues adhered to the tooth surface were removed using conical burs; this is because these substances could interfere with regeneration. The protocol device is exposed in extended mode in a 2021 publication [30]. No cuts to the periosteum were made to practice vestibular flap passivation because it was not necessary. The flaps were finally sutured with single stitches using a 3/0 silk thread. After about 7/10 days, the surgical site healing was assessed, and the sutures were removed (Figures 1 & 2).
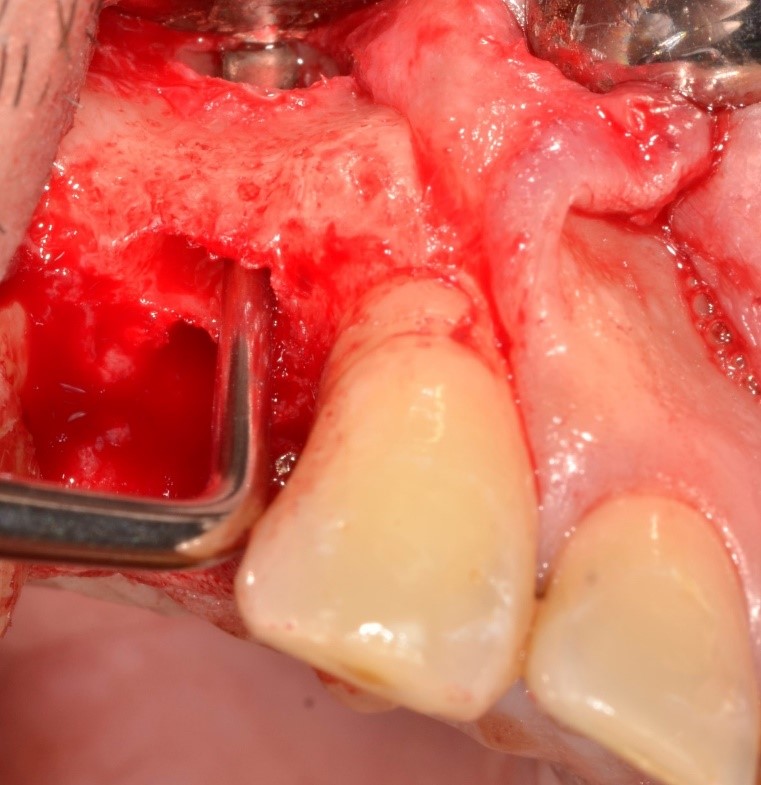
Figure 1: Intraoperative picture: after a full thickness flap, it’s possible to observe the bone defect, with an also buccal fenestration.
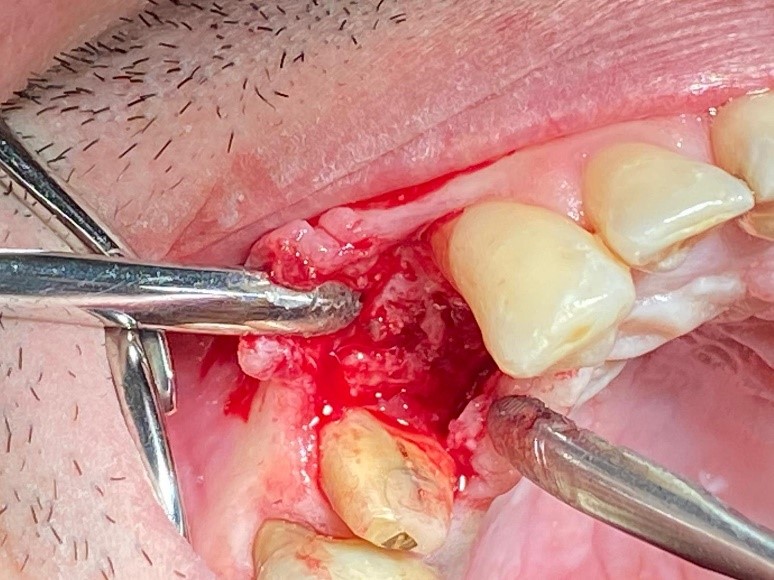
Figure 2: After three months, during the second surgery, it’s possible to appreciate regenerated bone volume to perform implant placement
Histological sampling and implant surgery
After three months, implant surgery is scheduled. After performing a cone beam CT scan, it was assessed whether bone regeneration had occurred successfully and whether sufficient bone tissue had been formed for one or more osseointegrated implants insertion. All the patients had an excellent recovery, without any type of complications, they are perfectly suitable for the second surgery phase. On surgery day, an antibiotic prophylaxis based on 2 grams of Amoxicillin and Clavulanic acid (875 mg + 125 mg) was administered. At first, a full thickness flap was per-formed to visualize the regenerated portion, which had a further clinical confirmation. Biopsies were performed using Trephine burs under copious irrigation of sterile physiological solution to cool the surgical site. Then the programmed implants were inserted. All patients did not present complications (Figures 3 & 4).
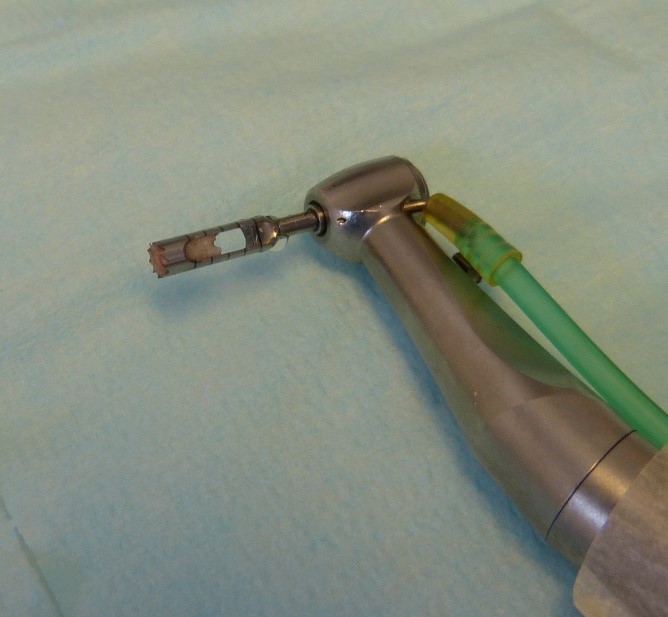
Figure 3: A biopsy sample inside Trephine.
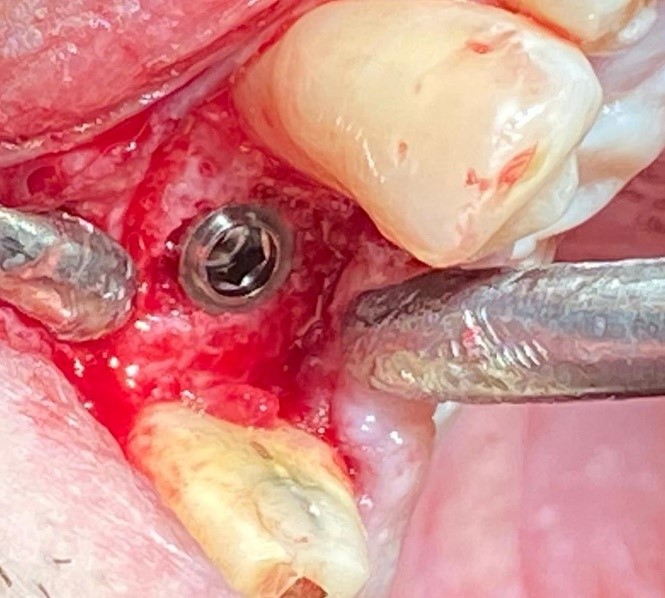
Figure 4: The implant was placed in the correct position with excellent primary stability.
Histological analysis
All samples were washed, dehydrated with alcohol solutions of increasing concentra-tion (Sigma-Aldrich, St.Louis, Missouri, USA), and then infiltrated into methacrylic resin ( Sigma-Aldrich, St.Louis, Missouri, USA) for the histological analysis. After, the sample was processed to obtain non-decalcified sections using disk abrasion system (LS2-Remet, Remet, Bologna, Italy) and diamond disk cutting system (Microm-et-Remet, Bologna, Italy) to obtained sample slides about 200 microns thick. Then, all samples were treated with low abrasive paper, on the lapping machine (Bueheler, Lake Blu_, Illinois USA) with thickness control allowing a progressive reduction of the sample thickness up to about 40–50 microns. Finally, the specimens were polished, colored with basic fuchsin and blue toluidine and observed with light and polarized light microscopy (Olympus, Shinjuku, Tokyo, Japan). The histological images obtained from the transmitted light microscope (Olympus, Shinjuku, Tokyo, Japan) were digitized through a digital camera and analyzed by means of an image analysis software IAS 2000 (QEA, Billerica, MA, USA). Each sample was split in nine sub sections. A per-centage of residual bone volume with exclusion of medullary tissues (BV%), a per-centage of the remaining graft, excluding bone and marrow (Graft%) and a vital bone percentage excluding the medulla and residual graft (VB%) were detected. Each sub section was measured using ImageJ program. In total, 909 sub sections were measured (Figures 5 & 6).
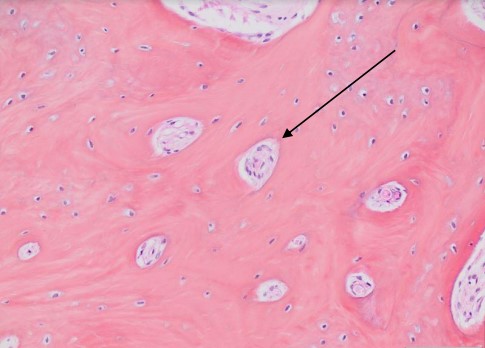
Figure 5: In this histological image it’s possible to observe the initial formation of the osteons and the characteristic circular arrangement of the bony lamellae is visible.
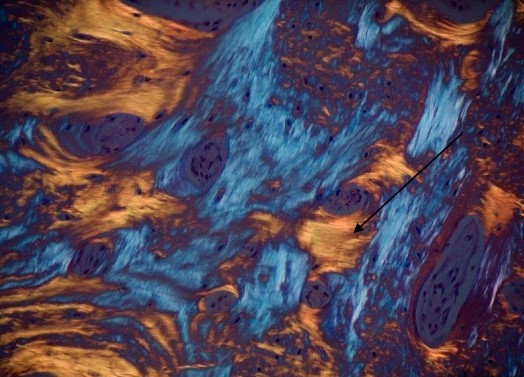
Figure 6: In this histological image, under polarized light, it is represented the direction of the lamellae which are arranged concentrically (yellow stripes).
Results
The patients did not report any problems in the postoperative days after surgery. Any pain was controlled with standard non-steroidal anti-inflammatory drugs and paracetamol. The removal of the sutures and constant checks showed a normal healing process. From the radiographic images, especially from CBCT, it was possible to detect a gain in bone volume in vertical and horizontal direction of the defects already present, and the preservation of bone volume where the post extraction sockets were present. Furthermore, both during the implant surgery and in slides of CBCT performed two months later it was possible to detect the presence of compact and dense bone tissue for primary stability. At the subsequent checks, no clinical signs of inflammation and pathological implant probes were detected. In the histological images, the presence of active osteoclasts on the surface of the dentin granules is relevant, now almost completely absent and small. Furthermore, it is possible to observe the apposition of newly formed bone which indicates an osteoblastic activity of tissue deposition and mineralization. There is no fibrous tissue between bone trabeculae. It is also possible to observe the presence of agglomerates of mesenchymal stem cells and the presence of vessels. No signs of tissue inflammation were detected (Figures 7 & 8).
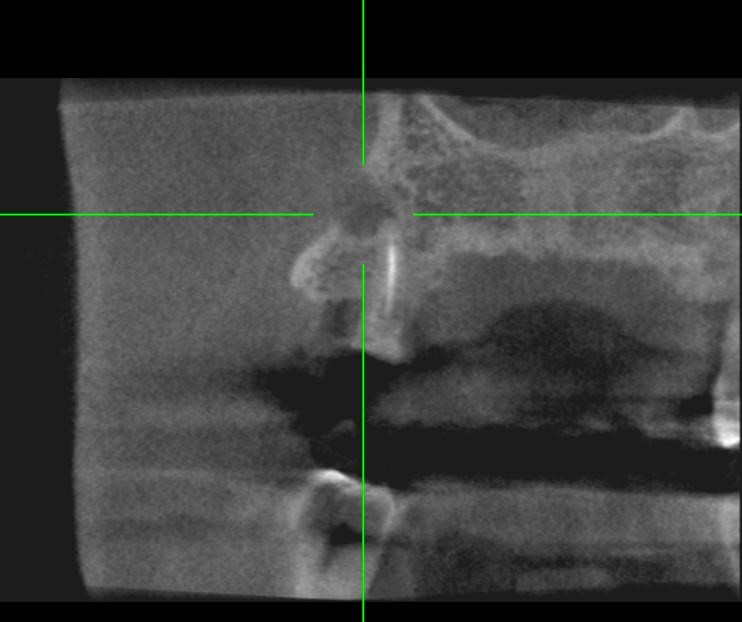
Figure 7: Pre-operative CBCT: this cross-section shows an infected root residue with its buccal fistula.
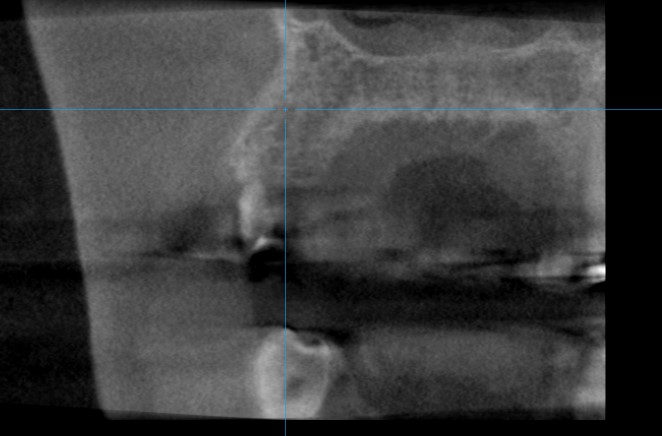
Figure 8: Pre implant surgery CBCT: three months after regeneration there is a sufficient volume of new bone tissue to place an implant.
Discussion
Many studies show that bone resorption, due to physio pathological processes follow-ing tooth extraction, is three-dimensional in height and thickness, and more extended in buccal area than in palatal/lingual area [19,20]. In the last 15 years, several surgical procedures, using autologous bone or heterologous biomaterials as bone graft, have been proposed to limit these processes [6,8]. However, in literature no trials showed only predictable benefits and responses. Today an extracted element is considered waste material [21-23]. The first aim of this study should prove that dental elements can be a more valid alternative than other graft materials in socket preservation procedure. The presence of bone tissue in the maturation phase, osteoblasts, osteoclasts and the almost total absence of dentinal granules is evidence of a successful re-placement of the graft and the addition of new bone tissue. The presence of numerous osteoclasts demonstrates how the organism has reacted to the graft and to the high concentration of growth factors present in it. Furthermore, the presence of agglomerates of mesenchymal cells, the presence of blood vessels and the absence of signs of inflammation show that this new tissue is vital and in a continuous phase of maturation; this is normal because the full maturation time of bone tissue is at least 6 months. The presence of small dentinal granules indicates that the graft derived from dentin is bio-compatible with a high replacement rate, as such as with other filling autologous material. It’s very important to know the timing of regeneration of the material because it allows to correctly plan the subsequent therapy phases, increasing the chances of success and decreasing complications. In clinical and radiographic checks, it was possible to appreciate an optimal bone tissue formation both from the volumetric and density. In the radiographic images it is possible to observe the tissue consisting of the outer cortical, vestibular and palatal/lingual, and of the inner medullary. No areas of thickening and resorption are present. Indeed, it was possible to insert an adequate number of implants to perform the rehabilitation with no need to perform further regenerative treatments. Despite the wait necessary for the formation of the material by the Tooth Transformer, it was possible to save time, money, and comorbidities [24,25].
Conclusion
The Tooth Transformer represents an interesting alternative in the field of tissue re-generation. In this study, the effectiveness of the material obtained with the device was evaluated, which brought numerous advantages and made the therapy more predictable, safer and cheaper. Among these advantages are the use of autologous material, bone regeneration, osteoinductive capacity of the material and gradual release of growth factors. All this is done ensuring simplicity and safety for the entire duration of the therapy, and without resorting to invasive samples but simply by using material deriving from the patient himself. In addition to this, the Tooth Transformer promotes bone regeneration, osteoinduction and wettability necessary for increasing osteoblastic maturation, for increasing the production of growth factors and for better mineralization of regenerated bone tissue. By viewing the histological images shown above, it can be seen how the grafted particulate not only the biocompatibility characteristics of common autologous materials has, but is also able to rapidly regenerate bone, ensuring an excellent result already three months after surgery.
References
- Weijden FVD, Dell’Acqua F, Slot DE (2009) Alveolar bone dimensional changes of post-extraction sockets in humans: A systematic review. J Clin Periodontol 36(12): 1048-1058.
- Schropp L (2004) Bone healing and soft tissue contour changes following single-tooth extraction: A clinical and radiographic 12-month prospective study. J Prosthet Dent 23(4): 313-323.
- Araújo MG, Silva CO, Misawa M, Sukekava F (2015) Alveolar socket healing: What can we learn? Periodontol 68(1): 122-134.
- Orgeas GV, Clementini M, Risi VD, Sanctis (2013) Surgical Techniques for Alveolar Socket Preservation: A Systematic Review. Int J Oral Maxillofac Implants 28(4): 1049-1061.
- Elio M, Martin C, Marcello C, Fabrizio C, Ugo G, et al. (202) Implants Survival Rate in Regenerated Sites with Innovative Graft Biomaterials: 1 Year Follow-Up 14(18): 5292.
- Mastrangelo F, Quaresima R, Grilli A, Tettamanti L, Vinci R, et al. (2013) A comparison of bovine bone and hydroxyapatite scaffolds during initial bone regeneration: An in vitro evaluation. Implant Dent 22(6): 613-622.
- Arx TV, Hardt N, Wallkamm B (1996) The TIME technique: A new technique for localized alveolar ridge augmentation prior to placement of dental implants. Int J Oral Maxillofac Implant 11(3): 387-394.
- Al Moraissi EA, Alkhutari AS, Abotaleb B, Altairi NH, Fabbro MD (2019) Do osteoconductive bone substitutes result in similar bone regeneration for maxillary sinus augmentation when compared to osteogenic and osteoinductive bone grafts? A systematic review and frequentist network meta-analysis. Int J Oral Maxillofac Surg 49(1): 107-120.
- Sanz M, Dahlin C, Apatzidou D, Artzi Z, Bozic D, et al. (2019) Biomaterials and regenerative technologies used in bone regeneration in the craniomaxillofacial region: Consensus report of group 2 of the 15th European Workshop on Periodontology on Bone Regeneration. J Clin Periodontol 21: 82-91.
- Pang KM, Um IW, Kim YK, Woo JM, Kim SM, et al. (2017) Autogenous demineralized dentin matrix from extracted tooth for the augmentation of alveolar bone defect: A prospective randomized clinical trial in comparison with anorganic bovine bone. Clin Oral Implant Res 28(7): 809-815.
- Kim YK, Kim SG, Yun PY, Yeo IS, Jin SC, et al. (2014) Autogenous teeth used for bone grafting: A comparison with traditional grafting materials. Oral Surg. Oral Med. Oral Pathol. Oral Radiol 117(1): 39-45.
- Kozuma W, Kon K, Kawakami S, Bobothike A, Iijima H, et al. (2019) Osteoconductive potential of a hydroxyapatite fiber material with magnesium: In vitro and in vivo studies. Dent Mater J 38(5): 771-778.
- Bang G, Urist MR (1967) Bone induction in excavation chambers in matrix of decalcified dentin. Arch Surg 94(6): 781-789.
- Gharpure AS, Bhatavadekar NB (2018) Clinical Efficacy of Tooth-Bone Graft: A Systematic Review and Risk of Bias Analysis of Randomized Control Trials and Observational Studies. Implant Dent 27: 119-134.
- Bessho K, Tanaka N, Matsumoto J, Tagawa T, Murata M (1991) Human dentin-matrix-derived bone morphogenetic protein. J Dent Res 70(3): 171-175.
- Rijal G, Shin HI (2017) Human tooth-derived biomaterial as a graft substitute for hard tissue regeneration. Regen Med 12(3): 263-273.
- Minetti E, Palermo A, Ferrante F, Schmitz JH, Ho HKL, et al. (2019) Autologous Tooth Graft after Endodontical Treated Used for Socket Preservation: A Multicenter Clinical Study. Appl Sci 9: 5396.
- Bono N, Tarsini P, Candiani G (2017) Demineralized dentin and enamel matrices as suitable substrates for bone regeneration. J Appl Biomater Funct Mater 15: 236-243.
- Kim YK, Kim SG, Oh JS, Jin SC, Son JS, et al. (2011) Analysis of the inorganic component of autogenous tooth bone graft material. J Nanosci Nanotechnol 11(8): 7442-7445.
- Mastrangelo F, Gastaldi G, Vinci R, Troiano G, Tettamanti L, et al. (2018) Immediate Postextractive Implants with and Without Bone Graft: 3-year Follow-up Results from a Multicenter Controlled Randomized Trial. Implant Dent 27(6): 638-645.
- Cardaropoli D, Nevins M, Schupbach P (2019) New Bone Formation Using an Extracted Tooth as a Biomaterial: A Case Report with Histologic Evidence. Int J Periodontics Restor Dent 39(2): 157-163.
- Grassi FR, Pappalettere C, Comite MD, Corsalini M, Mori G, et al. (2012) Effect of different irrigating solutions and endodontic sealers on bond strength of the dentin-post interface with and without defects. Int J Med Sci 9(8): 642-654.
- Comite MD, Crincoli V, Fatone L, Ballini A, Mori G, et al. (2015) Quantitative analysis of defects at the dentin-post space in endodontically treated teeth. Materials 8: 3268-3283.
- Minetti E, Berardini M, Trisi P (2019) A new tooth processing apparatus allowing to obtain dentin grafts for bone augmentation: The tooth transformer. Open Dent J 13: 6-14.
- Elio M, Andrea C, Marco C, Stefano C, Edoardo G, et al. Bone regeneration in implantology: tooth as a graft.
Citation: Carini F, Ferri L, Coppola G, Carini F, Monai D (2022) Derived Dentin Graft as Autologous Bone Regeneration in Implant Surgery: a Histological and Radiographic Investigation. Open Access J Dent Oral Surg 3: 1037

