Open Access Journal of Dental and Oral Surgery
[ ISSN : 2833-0994 ]
Immediate Implant Placement in Infected Sockets: Curettage Protocol Plus Disinfection with Hydrogen Peroxide and Rifamycin. Retrospective Analysis at 6 years of Follow-up
1Department of Implant and Biomaterial Research, Fundación Corazón de Jesús, San Juan, Argentina
2Research professor of Universidad Autónoma de Chile. Researcher of Instituto Murciano de Investigaciones
Biomédicas IMIB
Corresponding Authors
Keywords
Abstract
Context: Immediate implant placement in a fresh socket after extraction is an increasingly popular and established treatment option. However, active infection at the extraction site can adversely affect the outcome of this procedure.
Aims: This study was designed to evaluate the clinical results of immediate dental implant placement in infected extraction sockets.
Settings and Design: using a standardized protocol, which included the use of 3% hydrogen peroxide plus rifamycin 500 mg as irrigation for decontamination of the infected socket before implant insertion with submerged healing.
Patients and methods: The sample consisted of 227 implants placed in 158 patients, the data were obtained through a retrospective review of the clinical registry selected of patients who received implants according to the protocol described.
Statistical analysis used: A generalized binary response regression model was used. For all tests, the statistical significance value was set at p <0.05.
Results: Of the 227 implants placed immediately in the infected sites using the described protocol with an average follow-up of 6 years of prosthetic loading, only 11 failed, resulting in a cumulative implant survival rate of 95.2%. Only the medical history / harmful habits factor (HM / HH) was significant (p = 0.002), in this group the success rate was 85.7%. However, in the group of healthy patients without harmful habits, the success rate rises to 98.8%.
Conclusion: Immediate implant placement in infected sites using these protocols in healthy patients shows a survival rate similar to that published for implants placed in non-infected sites.
Introduction
Immediate placement of dental implants in fresh extraction sockets offers important advantages over early and late placement, such as minimizing the number of invasive surgeries and patient discomfort, as well as shortening the overall course of treatment. Early implant placement refers to implant placement after coverage and complete healing of the soft tissue of the extraction socket [1]. Success in animal and human studies has made immediate implant placement a routine treatment in dental practice [2]. The presence of active infection at the dental extraction site has been considered one of the main contraindications for immediate post-extraction implant insertion due to the risk of infection spreading to peri-implant tissues, leading to implant failure or retrograde peri-implantitis [3]. However, recent publications propose that immediate insertion of the implant into infected alveoli is not associated with a higher risk of failure compared to non-infected alveoli. It is currently emerging as a valid treatment option over more conservative two-stage approaches. Systematic reviews by Corbella et al [4] evaluated implant placement in infected extraction sockets. The human studies identified by Corbella et al reported survival rates ranging from 92% to 100% for a total of 497 implants placed at sites with endodontic infections with follow-ups of 12 months or longer. Fugazzotto [5] described a retrospective case series with 418 immediate implants placed in sites with periapical pathology. The implants were followed for an average of 67.3 months and the survival rate was 97.8%. Medikeri et al. [6] evaluated the effect of the concomitant use of platelet-rich fibrin (PRF) and decalcified lyophilized bone allografts (DFDBA) in immediate implants in extraction sockets with periapical infection, implant survival was 91.67% at 12 months.
The curettage and decontamination of the tissues of the infected alveoli to eliminate microbial remnants are the keys to success for the immediate placement of the dental implant. The literature reports the prescription of systemic antibiotics as a prophylactic measure to reduce the risks of infection [4]. In addition, it may be desirable to apply a local disinfectant to inactivate bacteria that reside in spaces inaccessible to mechanical cleaning. One method to surgically reduce the bacterial load is irrigation with antiseptic agents, which are superior to topical antibiotics since they are less selective in their action and are less likely to cause infection by resistant organisms [7]. Hydrogen peroxide-mediated bacterial death is believed to occur through multiple pathways, including DNA damage, as well as membrane protein and lipid oxidation [8]. In addition to direct bactericidal activity, multiple in vitro studies have shown that hydrogen peroxide can reduce biofilm formation by bacteria. Rifamycins inhibit the RNA polymerase of most bacterial genera and are used for the treatment of gram-positive prosthetic joint and valve infections, in which biofilms are prominent [9]. The importance of antiseptic synergy is that by combining agents, a broader range of organisms can be effectively covered and lower cytotoxic concentrations of individual compounds can be used [10].
Subjects and Methods
Informed consent for extraction and immediate dental implant insertion was obtained from all patients. The case histories of patients with implantation in fresh infected extraction sites were evaluated for this retrospective analysis by the dental surgeon in charge. One hundred and fifty-eight patients were identified with a follow-up average of 6,7 ± 3,9 years (mean ± standard deviation; range: 0-14 years) who received 227 implants according to the protocol described. One hundred fifty five implants were placed in the maxilla and 72 implants in the mandible. Treatment success was evaluated taking into account the factors of sex, age, jaw, arch area and medical history and / or harmful habits (osteoporosis, diabetes, collagen disease, bruxism, and tobacco smokers). All surgical procedures were performed by a single surgeon (SDRJ). Prosthodontic procedures were completed by (MAGV). The reasons for tooth extraction were classified, as suggested by Corbella et al [4] for each possible implant site as an endodontic cause, a periodontal cause, a root fracture, and an endodontic-periodontal cause or trauma combined with subsequent infection.
Informed consent was obtained for the extraction and immediate insertion of dental implants from all patients. All patients began with preoperative antibiotics 1-2 days before the surgical procedure Amoxicillin + clavulanic acid) (Optamox® 1gr-Roemmers, Argentina 1 gram twice a day, in the case of allergy to penicillin, ciprofloxacin (Ciriax ® Roemmers, Argentina) 500mg twice a day. The affected, fractured or infected root remains were gently extracted using minimally invasive techniques with a Pritchard’s periosteal (Hufrieddy® USA) and luxators (Premier® USA) to preserve the thin buccal bone wall. Extractions were performed without lifting flaps (Figure 1).
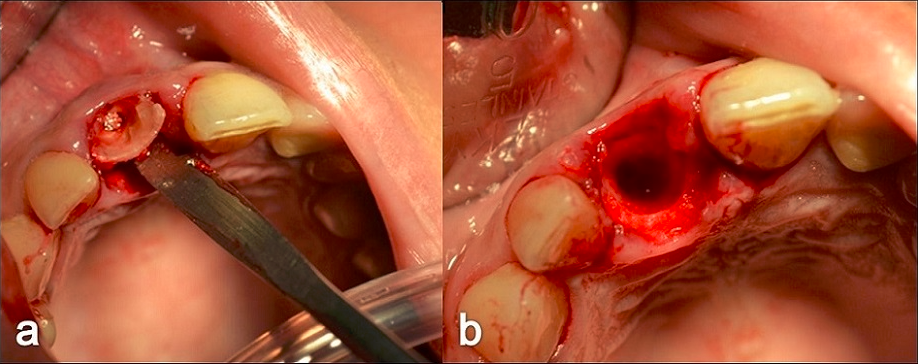
Figure 1: a) Delicate extraction of root debris with Pritchard’s periosteal. b) Postextraction socket with preserved buccal bone wall
Inflammatory tissues, pus, and necrotic tissues were removed by thorough thorough curettage of alveoli with Molt’s curettes, especially aggressive curettage was performed at sites where sources of infection were identified during tooth extraction. After a thorough mechanical cleaning, a key factor for the success of the protocol, the sockets were rinsed with sterile saline to remove tissue debris from the alveolus. Before preparing the implant bed, the sockets were irrigated with 3% hydrogen peroxide (10-volume hydrogen peroxide) for 60 seconds, (Figure 2) then rinsed with sterile saline to eliminate the remaining hydrogen peroxide retained in the cavity and avoid the consequent risk of air embolism [10].
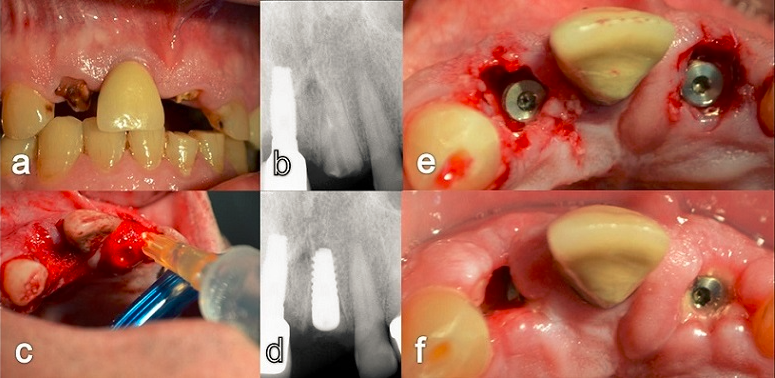
Figure 2: a) Preoperative b) Digital radiovisiography of the left central incisor with periapical infection c) Irrigation with hydrogen peroxide of the alveoli for 1 minute d) Digital radiovisiography of the immediate implant e) clinical vision f) healing at 12 days.
A final irrigation was performed with rifamycin SV 500mg local use solution Rifocina ®, Argentina for 60 seconds prior to drilling or preparation of the bed for immediate insertion of the implant in the socket. This procedure was repeated twice for each alveolar cavity. The extraction sockets were considered suitable for immediate implant insertion provided that initial implant anchorage could be achieved even if the vestibular plate was affected. The alveolar graft was performed with beta [11] tricalcium phosphate + Membracel® Argentina collagen membrane (crushed), achieving a biomaterial with a macillo-like consistency to fill bone defects caused by pre-existing inflammatory processes or gaps greater than 3 millimeters. The material was filled to bone level, covering the cover screw in most cases. The bone graft substitute used has the inherent ability to harden in contact with blood, thus avoiding the need for a membrane for graft retention. The defect was sutured using a 3-0 silk suture (Ethicon®, Johnson & Johnson Ltd.) although primary wound closure was never achieved as no flaps were performed. In addition, whenever possible, implants were placed 1 to 2 mm subcrestally to optimize the platform change functions of the implant system.
The implants were loaded 143 ± 39 days (mean ± standard deviation) after implant placement (Figure 3 cdef). Statistical analysis used. The success / failure rate of the treatments according to the various categorical factors (sex, jaw, etc.) was described using their respective relative and absolute frequencies, while for the scalar parameters (age, loading time and follow-up time) these were described by means and standard deviation. To determine the magnitude of the effects and significance of the aforementioned factors on the treatment outcome (success / failure), a generalized binary response regression model was used. For all tests, the statistical significance value was set at p <0.05.
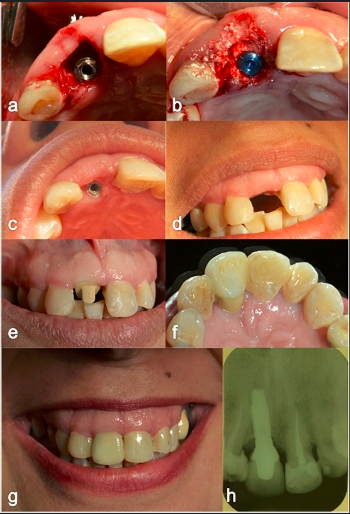
Figure 3: a) Immediate post-extraction implantation with GAP greater than 3 mm. b) Filling the GAP with beta-tricalcium phosphate and crushed collagen membrane. c) Clinical view of the palatal position of the implant. d) Frontal view e) Personalized ceramic abutment f) Palatal view of the completed crown. g) Clinical view of follow up at 7 years and h) Periapical X-ray, maintenance of the crestal bone level is observed.
Results
Number of patients: 158; Implants placed: 227 Distribution of patients according to sex: Most of the patients were women (n = 100; 63.3%), almost twice as many as male patients (n = 58; 36.7%).
Success / failure rate according to gender
One hundred and forty-four implants were placed in women, of which only 5 (3.5%) failed, while 83 implants were placed in men and 6 (7.2%) failed. The overall success rate was 95.2%, and it was higher in women (96.5%) than in men (92.8%).
Relationship between the success / failure of the implant and the age of the patients
Although the age factor showed an increasing trend (the older the age, the greater the probability of failure), the effect was not significant (p = 0.15).
Success / failure rate according to jaws
The success rate was higher for implants placed in the maxilla (96.1%), in contrast to mandibular implants (93.1% success).
Success / failure rate according to sector
None of the 30 implants placed in the molar sector failed (100% success). On the other hand, the sector with the highest risk of failure was premolars, of the 107 implants placed in this sector, 7 failed (6.9%). Regarding the incisor and canine sector, the success rate was 95.7% and 96% respectively.
Success / failure rate according to medical history and / or harmful habits (MH/HH)
The difference between the groups was noticeable; the success rate of implants placed in healthy patients without harmful habits (bruxism and / or smoking) reached 98.8%, while in the other group of patients with HM / HH success rate was 85.7%. The differences were significant (p <0.05).
The average age was higher in patients with failed implants. Figure 4 show relative frequencies of implant success and failure according to: a) Gender, b) Age c) Jaws, d) Sector and e) Medical history and / or harmful habits (MH / HH).
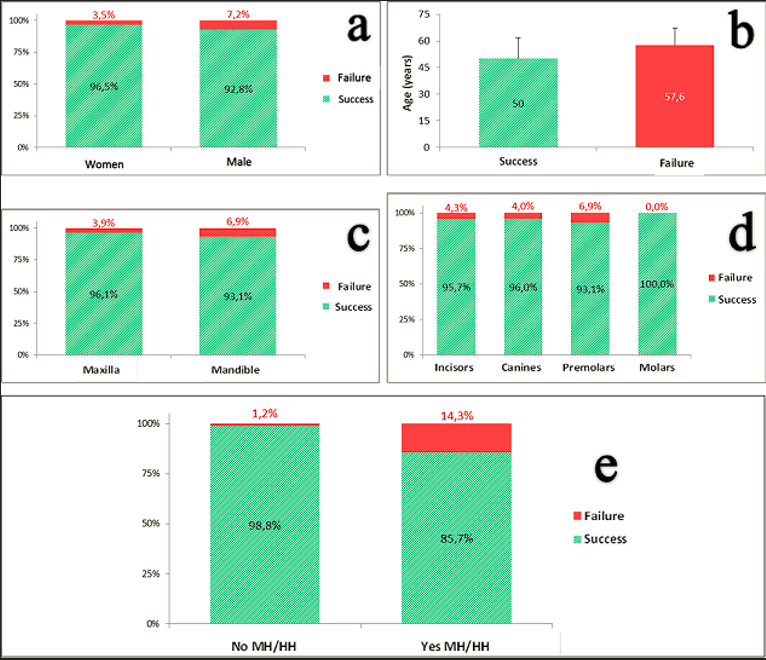
Figure 4: Relative frequencies of implant success and failure according to: a) Gender, b) Age c) Jaws, d) Sector and e) Medical history and / or harmful habits.
Treatment follow-up time
The treatment follow-up time was 6.7 ± 3.9 years (mean ± standard deviation), within a range of 0 to 14 years. The vast majority of failures were prior to implant loading (n = 8) and only three cases failed, two a year after placement and the other within a year.
Implant loading time
The implant loading time was 143.5 ± 39.2 days (mean ± standard deviation), within a range of 0 to 210 days (immediate loading was performed on 8 implants).
Analysis using generalized linear regression model (GLRM)
To determine the magnitude of the effects and significance of the aforementioned factors on the treatment outcome (success / failure), a generalized binary response regression model was used.
Response variable: treatment result (success / failure)
According to the results obtained, the factor “medical history/ harmful habits” verifying a significant effect in relation to the success or failure of the treatment which can observe in Table 1.
Table 1: Result of the binary response generalized linear regression analysis: Origin of the effect; Observation on the conditions associated with an increased risk of implant failure; Wald Chi2 statistic and significance of the effect (p-value).
|
Origin of the effect |
Higher probability of failure |
Chi2 de Wald |
p-value |
|
MH / HH |
With medical history and / or harmful habits |
9,27 |
0,002 |
|
Age |
Older Patients |
2,03 |
0,15 |
|
Jaws |
Mandible |
1,12 |
0,29 |
|
Sector |
Premolars |
0,83 |
0,84 |
|
Gender |
Male |
0,01 |
0,92 |
The effects of the other factors evaluated were not significant, although in relation to the age of the patient, the observed trend was that the older the age, the greater the probability of failure.
Most of the implants that were followed showed normal peri-implant mucosa and no visible plaque (Figure 3g).
Regarding complications, two fractures of the ceramic phase of the dental crown were reported in a patient who developed bruxism and was not used to the use of the indicated relaxation plate. No other biological or mechanical complications were reported.
Discussion
In our study, the results of the protocol were satisfactory and the cumulative implant survival (95%) is in the range reported by the systematic review by Corbella et al [4], which ranged from 92% to 100%. However, a systematic review and meta-analysis by Oliveira-Neto et al [12] reports that immediate dental implants placed in infected sites present a higher risk of failure than immediate dental implants placed in non-infected sites. Furthermore, an evaluation of dental implant insertion protocols by Tettamanti et al. [13] concluded that there is insufficient evidence in the literature to determine the possible advantages or disadvantages of immediate insertion, early insertion, or delayed insertion of the implant. These studies suggest that immediate and early insertion may have a higher risk of implant failure and complications than delayed implants; However, they consider that the aesthetic result may be better when placing the implants just after the extraction of the teeth.
Currently the decision of the protocol to follow is controversial, it is known that a delay in the insertion of the implant translates into a loss of available bone volume. Ahmed [14] states that in the first seven days after extraction, the greatest dimensional changes occur in the alveolar ridge with the mean maximum negative deviation, identified by 3D analysis, a bone loss of -1.7 mm ± 0 was recorded. , 3, compared to day 0 and the mean 3D reduction in alveolar ridge width was -3 mm at four months compared to one week data. The highest rate of dimensional changes occurred within the first seven and twenty-eight days after extraction. This inexorable loss of bone volume that occurs in the first month after tooth extraction will impact the functional and aesthetic result. Numerous studies have shown that after tooth extraction, approximately 30% of the alveolar ridge is lost as a result of resorption [15].
Conclusion
Immediate implant placement in infected alveoli is promoted with a meticulous curettage protocol plus disinfection. Using 3% hydrogen peroxide plus rifamycin 500 mg solution for local use as irrigation for the decontamination of the infected alveolus before implant insertion shows a survival rate (98.8%) similar to published success rates for implants placed in non-infected sites in healthy patients. However, patients with “medical history / unhealthy habits” represent a significant factor in the success of the treatment that decreases to (85.7%). Therefore, we consider that the benefit of preserving alveolar bone supports the use of this disinfection protocol only in healthy patients.
References
6. S Medikeri R, Meharwade V, M Wate P, V Lele S (2018) Effect of PRF and Allograft Use on Immediate Implants at Extraction Sockets with Periapical Infection -Clinical and Cone Beam CT Findings. Bull Tokyo Dent Coll 59(2): 97-109.

